Plasma Transfusion Practice & Antibody Pageants and Parades
Presented by: Dr. Nadia Gabarin & Neeru Sahni
Evaluation Survey:
Link: https://orbcon.limesurvey.net/157514?lang=en
QR Code:

Attachment(s)
Donna Berta, RN, BScN
Access Pre & Post Transfusion Knowledge Questions and Answers with Rationale here
Attachment(s)
https://transfusionontario.org/wp-content/uploads/2026/03/Transfusionists-Talk-2026-Mar_final.pdfWritten by:
Akash Gupta, MD FRCPC DRCPSC CTBS Transfusion Medicine Specialist, Precision Diagnostics and Therapeutics Program (Laboratory Medicine), Sunnybrook Health Sciences Centre , Medical Officer, Canadian Blood Services
At the surface, the administration of blood products requires that we issue the right product to the right patient at the right time. However, behind this apparent simplicity, there is a complex series of events where errors can occur, including collection and production with the blood manufacturer, patient testing and issuing at the blood bank, and ordering and administration with the clinical team. To catch these mistakes and correct the systematic issues that cause them, we rely on our hemovigilance systems. In Canada, that role is filled by the Transfusion Error Surveillance System (TESS), a national program that plays a crucial role in transfusion safety.
TESS was launched in 2005 by the Public Health Agency of Canada (PHAC), as a result of the Krever Commission (X). The commission enforced the idea that the safety of our blood supply was not based solely on testing of the products, but also monitoring how transfusions are ordered, prepared, and administered. The program started with 11 hospitals and by 2008 had evolved into a national surveillance system with 15 hospitals in 4 Canadian provinces and territories acting as sentinel sites and reporting all errors identified. The involved sites account for approximately 16% of the blood transfusion activities in Canada.
TESS collects non-nominal, standardized data on transfusions errors and classifies them as near misses (not reaching the patient) or actual events, which are further classified as causing harm or no harm. The 2012-2016 TESS report summarized 50,000 errors, with 95% classified as near-miss events and 93% of those as planned discovery events. A more recent report from 2022, summarizing approximately 10,000 errors, had 92% of errors classified as near miss and only 3 events (0.4%) causing patient harm. In addition to confirming the overall safety of blood transfusion in Canada, the aggregate data from these reports is helpful in identifying sources of potential errors or contributing factors to repeated near-miss events. For example, the 2012-2016 TESS report highlights that the greatest number of errors occur with sample collection (34% of all errors reported).
The planned sunsetting of the Transfusion Error Surveillance System represents a significant transition point for transfusion safety in Canada. Over nearly two decades, TESS has provided a national lens on transfusion errors, enabling hospitals to benchmark performance, identify systemic vulnerabilities, and demonstrate that most errors are intercepted before patient harm occurs. The loss of this coordinated national surveillance framework risks creating gaps in our collective understanding of error patterns and trends, particularly for near misses that rarely surface through adverse reaction reporting alone. As Canada moves forward, sustaining the gains achieved through TESS will require renewed commitment to local error reporting, strong hospital-based hemovigilance programs, and consideration of alternative national or provincial mechanisms to ensure continued learning, transparency, and improvement in transfusion practice. Without such efforts, the quiet safety successes fostered by TESS may become harder to measure—and harder to maintain.
Attachment(s)
Written by:
Dr. Andrew Shih TTISS Ontario Director
Melanie St John TTISS Ontario Coordinator
The Transfusion Transmitted Injuries Surveillance System (TTISS) continues to play a crucial role in safeguarding the health of Canadians by monitoring and reporting adverse reactions to blood products, including blood components and plasma derivatives. The commitment of all 159 Ontario hospitals contributes to the continuous improvement and effectiveness of TTISS.
On November 24th, 2025, TTISS Ontario hosted a highly successful hybrid meeting, attracting over 200 attendees. This event provided a platform for all to share insights, discuss challenges, and collaborate on enhancing transfusion safety. Attendees had the opportunity to hear from several outstanding speakers during the event, with topics including: adverse reaction reporting in rural and community settings, an update for the revision of the TTISS Manual, pathogen reduction and its relevance to adverse reactions, interactive case presentations, and transfusion reactions in the operating room.
A major initiative for 2025 is the comprehensive update of the TTISS Manual, which has remained unchanged since 2007. This revision aims to incorporate the latest advancements in adverse reaction definitions highly informed by the recent Quebec INSPQ version of the manual for alignment and guidance from a multidisciplinary stakeholder group; to inform future best practices.
TTISS Ontario also showcased its work at the most recent Canadian Society for Transfusion Medicine (CSTM) conference in Newfoundland, presenting two posters: one examining the impact on pathogen-reduced platelet implementation, and another exploring the Impact on solvent detergent plasma on adverse transfusion reaction reporting. Hemovigilance and adverse reactions will be featured in multiple sessions/workshops at the 2026 conference held in Hamilton, Ontario.
Finally, with the sunsetting of the Blood Safety Contribution Program in April 2026, a consensus conference is currently being planned to guide recommendations for the future of hemovigilance. Interviews and stakeholder engagement have begun to inform the proceedings. PHAC has also confirmed that access to the Canadian Network for Public Health Intelligence (CNPHI), a web-based data platform, will remain available to BSCP recipients until March 31, 2027, allowing provinces and territories to continue submitting data. This extension will serve as a bridge while a long-term hemovigilance framework is under development.
If interested, please visit our TTISS Website for additional resources.
Attachment(s)
Written by:
Laura Aseltine MLT, BSc., Regional Project Coordinator, Southwest;
Donna Berta RN, BScN, Clinical Project Coordinator – Nursing;
Sheena Scheuermann MLT, BTech., Regional Project Coordinator, Southwest;
Alison Wendt MLT, Regional Project Coordinator, Central.
Background
ORBCoN’s Transfusion Medicine Boot Camp for Nurses is an annual educational event (originating in 2018). The goal is to enhance transfusion patient safety by providing evidence informed transfusion knowledge and skills to health care professionals who administer blood (primarily nurses). In addition, the perspective of medical laboratory technologists, whose role is integral to the transfusion medicine circle of care, is embraced.
The program’s organizing committee includes a transfusion medicine physician, clinical nurse educators, a patient blood management coordinator, and ORBCoN staff. Program format and content are guided by attendee feedback.
2025 Program
A four-hour virtual educational session was presented on November 26, 2025. Registration to attend was required (for security purposes) however there was no cost to attendees. The 2025 format was modified to include four consecutive sessions; based on attendee feedback and limitations of the available technology, the concurrent session format from previous years was abandoned. The 2025 contemporary transfusion medicine topics included:
- Transfusion Considerations – Patients with Liver Disease
- Transfusion Considerations for Obstetrical Patients
- Tips for Transfusing Tots
- 2025 Using Blood Wisely Guidelines: Are You Transfusing Wisely?
For each topic, the recording, presentation (PDF format), and as applicable, speaker responses to additional questions are posted on the ORBCoN website.
Visit https://transfusionontario.org/category/orbcon-resources/presentation-library/transfusion-medicine-boot-camp-for-nurses/ to access these resources.
2025 Registrants
Those registering for the event totaled 363 individuals; most registrants were Ontarians. Approximately 6.6% of registrants were from outside Ontario, including two international registrants.
The role of the registrants is summarized in Figure 1, details include:
- 51% (N 186) front line nursing care provider (clinical nurse specialist, registered nurse, registered practical nurse)
- 27% (N 96) providing transfusion education (clinical nurse educator, nursing professional practice, transfusion safety nurse/officer)
- 14% (N 51) medical laboratory technologist
- 8% (N 30) other (faculty university/college, nurse manager, student nurse, other)
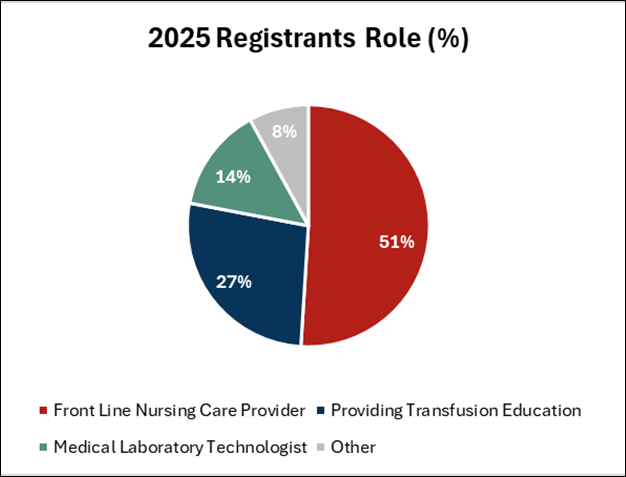
Figure 1: 2025 Registrants Role (%)
2025 Outcomes
An evaluation survey provided attendees the opportunity to submit feedback and was completed by 82% of attendees (150 completed surveys, 182 attendees). Common responses the attendees identified:
- Overall appraisal: 77% excellent, 23% good.
- After attending this event, my practice will change: 83% yes.
- The varied topics were well liked and described as “informative” and delivering “practical knowledge for nurses.”
- The 2025 event four-hour duration: 81% adequate, 13% too long, 6% too short
- Program organization with 50 minutes per speaker and a 10-minute break scheduled between speakers was positive; attendees reported selecting the specific topic(s) most pertinent to their individual clinical practice focus.
- Attendees remarked that the content of some presentations did not align within the allocated time period leading to minimal/no question and answer discussion (subsequently the speakers were asked to address attendee questions in written format, with replies posted on the ORBCoN website). Attendees clearly requested question and answer discussion opportunities for future events.
- Most attendees found the interactive polling questions engaging, though some voiced concern, “too many polling questions.”
- A polling question technology malfunction arose for many attendees; moving forward ORBCoN is investigating to rectify this issue.
- Patient case scenarios were favorably received with requests for “nursing patient cases.”
- Suggested future topics: Artificial Intelligence in Transfusion; Patient Case Demonstrating Transfusion Protocols; Leukemia and Transfusion; Antibodies and Crossmatch Process; Operating Room Transfusion Protocols; Dialysis Patients and Transfusion.
Planning for the 2026 Event
Stemming from the 2025 event, as well as past years feedback, education to support transfusion patient safety is welcomed. Attendee observations and suggestions are valued; planning suitable responses are a priority. As possible, the future topics put forward will be incorporated.
Your ongoing suggestions to augment this event are encouraged, please reach out.
Attachment(s)
Written by:
Keescha Wherry, Clinical Educator, RN Ellen Wiley, Clinical Educator, RN
Transfusing blood and blood products can be a daunting task for a novice practitioner. This is a complex and high-risk process that is life saving and life enhancing; but conversely can lead to negative outcomes. The team must ensure there is a clear evidence-based rationale for any transfusion, and the product is prepared, tested, and dispensed following stringent guidelines. Then the patient can be transfused in a manner based on protocol to enhance patient safety.
In 2021 we created our own training modules for our nursing staff. We recognized the importance of refreshing knowledge and sharing updates in practice. We created a module to complete upon hire, and a short annual refresher which is reviewed and launched yearly. In preparation, we began attending Transfusionists Talks to ensure our knowledge was accurate. We also focused on the physician group- training in a conservative approach to align with best practice.
We see first-hand the critical role blood products play in patient health. In 2022 we launched our hospital-wide Code Transfusion Policy. Not long after launch we experienced our first need for a massive transfusion protocol- an obstetrical postpartum haemorrhage (PPH). PPH remains one of the leading causes of morbidity and mortality (Robinson et al., 2022) in maternity care, and the timely administration of blood products can mean the difference between life and death. Our Code Transfusion policy standardized our rapid response to massive haemorrhage, including obstetrical haemorrhage. This standardization ensures clear communication, collaboration, and response, which in turn improves confidence and clarity.
In 2025, our team proudly attained the “Choosing Blood Wisely” designation: an achievement that reflects our strong commitment to clinically validated transfusion practices. A key component of this process has been the use of Transfusionists Talks, including participation by laboratory staff, front-line nursing staff, staff development, and physicians. The Transfusionists Talks were instrumental in the development of our practical and research-driven protocols: including blood management principles and strategies to ensure minimal blood exposure while maximizing access when the acuity of the situation demands it.
Transfusionists Talks also helped bridge the divide between disciplines: physicians, nurses, and laboratory staff were all given the same messaging. This improved practices, documentation, processes, and heightened awareness of blood stewardship. Transfusionist Talks reinforced that transfusion is not just a clinical act, but a decision with long-term implications.
Achieving the “Choosing Blood Wisely” designation is more than just a badge of honour: it is a testament to our institution’s commitment to continued improvement, and a conscientious effort to enhance patient safety, optimize resources, and cement our cohesiveness.
Our journey demonstrates that education, collaboration, and evidence-based protocols are powerful tools for change. As we look ahead, we remain committed to an evidence-based approach to blood management throughout the organization.
Reference:
Robinson, Debbie, Basso, M., Chan, C., Duckitt, K., & Lett, R. (2022). Guideline no. 431: Postpartum hemorrhage and hemorrhagic shock. Journal of Obstetrics and
Gynaecology Canada, 44(12),1293–1310. https://doi.org/10.1016/j.jogc.2022.10.002
Attachment(s)
Written by: Alexis iob – ORBCoN Regional Project Coordinator & Tracy Cameron- ORBCoN Regional Manager
ORBCoN offers a comprehensive range of resources to support best practices in Transfusion Medicine. Our goal is to ensure healthcare professionals have access to up-to-date information through regularly updated documents and tools.

Accessing Resources
When you visit the transfusionontario.org website and click on the “Resources” tab it will open a screen with a blue box where you can view the different categories available to you. If you know exactly what you are looking for, you can always use the search function at the top right-hand side of the screen.
Resource Categories

Bloody Easy E-tools & Publications: Register for the E-learning modules such as ORBCoN Tech Assess and upcoming Reconstitution Outside the Transfusion Medicine laboratory (TML). Access flipbooks and PDF handbooks for Bloody Easy Blood Administration (BEBA), Bloody Easy 5.1, and Bloody Easy Coagulation Simplified. Physical copies can be ordered online. Ontario orders are free; out of province orders incur fees. To order the handbooks you will fill out a form and they will be shipped to you.

Blood Utilization Graphs & Reports: Find Provincial audit tools like the Bedside Audit of Blood Administration that hospitals can use as part of their quality improvement initiative project. Annual blood utilization information for Ontario can also be found in this section, along with other provincial reports like the Canadian Obstetrical and Perinatal Transfusion Network (COPTN) national survey report and other audit reports conducted in previous years.

IVIG/SCIG: This section features the Ontario Utilization Management Guidelines, Fact sheets for patients, how to order IG in Ontario, and the IG dose calculator. If your site is interested in gathering hospital specific IG utilization data, there is a link to register for the IG quality improvement project.

Massive Hemorrhage Protocol (MHP): Comprehensive resources for managing critical bleeding scenarios. Learn about the Ontario MHP and access companion tools such as eLearning modules, simulations, checklists and algorithms. Use the provincial MHP toolkit to aid in implementation in your hospital and to track and monitor quality metrics using the MHP quality metrics portal and dashboard.
Toolkits: The Toolkits collection is designed to support Transfusion Medicine practitioners with practical, scenario-specific resources—ranging from audit checklists to emergency protocols, all tailored for use at the point of care.
| Toolkit Name | Key Feature |
| Bedside Audit | Tools to support standardized bedside safety checks and practice audits |
| Dispensary & Administration | Guidance on secure and compliant blood product dispensing |
| Emergency Blood Management | Resources to prepare and respond to acute blood shortages or massive hemorrhage events |
| Home Infusion Toolkit | Practical materials for delivering transfusions safely outside institutional settings |
| Lab Evaluation Process | Tools to implement and evaluate a Transfusion Medicine Laboratory’s performance |
| Competency Assessment Program (CAP) | Templates for verifying staff competence—technologists, nurses, physicians |
| Inventory Management | Best-practice guidance on stock control and ordering processes and redistribution |
| New Product | Frameworks to facilitate introduction of new blood products |
| Resources for Midwives | Specialized content for prenatal, perinatal, and neonatal transfusion planning |
| Special Blood Requirements | Guidance on managing rare or patient-specific blood needs |
| Internal Blood Transport | Protocols to ensure traceability and safety during product movement within facilities |
| Transfusion Committee Toolkit | Materials for administering committee responsibilities like policy review and governance |
| Transfusion Safety Officer Resource Manual | Checklists and tools to support this pivotal role |
| Quality Improvement Plan | Templates and guidance to support audit-driven quality initiatives |
| OTTRM | We are currently revising this resource. If you are looking for a specific procedure complete the request form on the web page. |

ORBCoN Resource: The ORBCoN Resources page is an essential access hub for transfusion professionals—offering access to past educational materials, previous ORBCoN report newsletters, helpful apps related to transfusion medicine, and more. Content is organized under main tabs: Presentation Library, Helpful Apps, Order Resources.

TM Guidelines & Standards: These resources are designed for transfusion medicine leaders, safety officers, and clinical teams to ensure evidence-based care and regulatory compliance. A comprehensive set of resources includes clinical guidelines for blood components and specialty areas, national recommendations from the NAC, and practical tools like bleeding management apps and transfusion safety tips. The page also offers regulatory standards, accreditation guidance, and a detailed resource manual for medical directors to support governance and quality assurance.

Quality Improvement: This resource library is currently under revision, but will showcase the Ontario Transfusion Quality Improvement Plan (OTQIP) along with a section dedicated to Health Canada and Accreditation Canada Diagnostics information to help hospitals meet the regulations and requirements set for Transfusion Medicine.
We greatly appreciate your support for ORBCoN and if you have any questions or ideas for new resources, please reach out to transfusionontario@gmail.com.
Attachment(s)
Written By:
Andrea Tjahja, MLT, ART, BSc, BEd Vice President, Operations Medical Laboratory Professionals’ Association of Ontario
The Medical Laboratory Professionals’ Association of Ontario (MLPAO) is the fastest growing lab association in Canada. Founded in 1963 as the Ontario Society for Medical Laboratory Technologists (OSMT), the MLPAO’s membership has increased by over 73% in the past 5 years to more than 4,200 members across the province, including both MLTs and MLA/Ts.
MLPAO is your voice in Ontario. The MLPAO advocates for the lab profession, monitoring legislative changes and engaging with the Ministry of Health, employers, and other stakeholders. Over the past 5 years, our advocacy efforts have resulted in historic funding of over $15M for lab professionals including 700 more seats for MLTs and MRTs and inclusion in the Learn and Stay Grant, which provides free education in the MLT programs at St. Clair College and Cambrian College. We continue to work for you to address the MLT shortage and other issues affecting the lab profession.
MLPAO members receive professional liability insurance (PLI) included in their membership. PLI coverage also includes professional legal and HR advice as well as data security and privacy breach consulting services. Our PLI covers MLT members across Canada. There is no extra cost for PLI – that’s a little extra that stays in your pocket!
Continuing education (CE) is top of mind at MLPAO. Members have access to our free monthly Lab Talks webinars, hosted by laboratory experts from across the province – our members love them! Our online CE course offerings are current, continuously updated throughout the year, and provide you with a certificate for your professional portfolio. We are also excited to announce our 2026 Lab Summit – Together in Toronto on June 5-6th in Toronto. We invite you to join us for a day of connection, collaboration and community.
Membership with the MLPAO gives MLTs and MLA/Ts professional liability insurance (PLI), continuing education (CE), advocacy, and so much more. Stay covered, stay current, and stay connected with your professional community.
Learn more at mlpao.org.
Attachment(s)

We invite you to listen to “The Inflection Point Podcast” where Dr. Kimmo Murto and Dr. Jeannie Callum address pediatric trauma, providing practical insights for paramedics, physicians, and trauma teams managing pediatric emergencies including pediatric massive hemorrhage protocol.
The Inflection Point is a Canadian paramedic podcast dedicated to advancing EMS education, prehospital care, and healthcare leadership. Hosted by Jakob Rodger and Ryan Cichowski, the show brings evidence-based discussions on pathophysiology, pharmacology, trauma, cardiac care, human factors, and clinical decision-making.
Attachment(s)
Written by: Sheena Scheuermann – ORBCoN Regional Project Coordinator

The mission of GHEST is to promote education, research, and best practices in Transfusion Medicine throughout the Golden Horseshoe region. The model is intended to be a non-profit venture therefore the symposiums are held at minimal cost to participants.
This year’s event was held in Oakville, Ontario. The symposium was well attended with 119 registrants for the day. We were very fortunate to have a patient speaker, Jules Royer who shared her journey with Multiple Myeloma. Attendees consistently express how impactful it is to have a patient speaker start the day.
Following this, Dr. Danyal Ladha presented an Introduction to Plasma Cell Dyscrasias, which provided the audience with information regarding diagnosis, treatment and insights into new advances in treatment.
Rebecca Barty introduced the new Perinatal Recommendation statements, which followed an open discussion with the attendees on potential challenges laboratories may see with implementing the new recommendations. We thank the attendees for their participation in the discussion as it provided useful insights into the challenges and possible tools that can be developed to assist with implementation.
The afternoon consisted of presentations by Dr. Sheharyar Raza on AI applications for blood bank, and Tracy Cameron on Blood Administration Bedside Audit followed with a live simulation of a transfusion lead by Laura Aseltine and Donna Berta. The audience was encouraged to participate and instructed to point out the errors in the process as they arose during the simulation. The feedback regarding this type of activity was positive and we look forward to doing something like this in the future.
The day was rounded out with a case study presentation regarding a patient with sickle cell anemia by Dr. Jordan Radigan. Including more case studies in the program continues to be a suggestion from attendees.
Thank you to our Sponsors CSL Behring, Grifols, Octapharma, CSTM and ORBCoN! Once again, a huge thank you to the speakers and organizing committee for another successful GHEST event.
Presentations will be posted on transfusionontario.org.